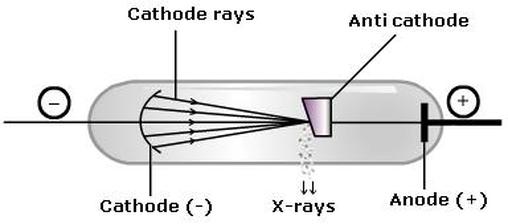
 The nature of X-rays: * X-rays are Electromagnetic waves. * Highly energetic, short wavelength (10^-8m - 10^-13m) How are X-rays produced ? Follow the diagram on the left. The electrons are produced at the hot filament cathode. These electrons are accelerated towards the anode (tungsten) by applying a high voltage between the electrodes. The fast moving electrons are then decelerated as it hits the tungsten. In these process the kinetic energy of the electrons is transformed into the x-ray photons. About 1% of the electrons energy is converted into x-rays, rest energy is transferred to tungsten heating it up therefore to cool down the tungsten, water is circulated through it.
0 Comments
The photoelectric effect was first proposed by a patent clerk Albert Einstein who later won the noble prize for this paper and later became famous for the theory of relativity.
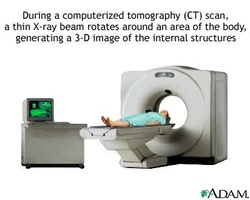 How modern CAT scan works? A patient is placed in a ring of X- ray detector. X-ray beam passes through the patient at different angles. A thin fan shaped beam is used Detectors opposite to the x-ray tube measures the intensity if x- rays absorbed by the patient. This detectors are all connected to the computer. Images of slices through the patient in one plane is produced using computer software. X- ray is moved along the patient for the next slice through the patient. Advantages of using CAT: Produce 3D image. Better constrast between different soft tissuses. Shows precise position and shape of tumors . They can taken quickly so a large number of patients can be scanned per day. Difference between A- scan and B- scan: A- scan is one direction B- scan uses number of sensors at differenct angle to build up 2D or 3D The advantage of using MRI scan is that it does not involve radioactive source so no risk. The nuclei of an atom have a property called spin. Spin is the intrinsic angular momentum of particles.
Until the early 20th century, physicists thought that the atom was a positively charged globule with negatively charged electrons sprinkled in it. This "plum pudding" model was called thomson model as it was proposed by J. J. Thomson, who discovered the electron in 1897, was proposed in 1904 before the discovery of the atomic nucleus in order to add the electron to the atomic model. This all changed in 1909 when the Rutherford scattering experiment was done. 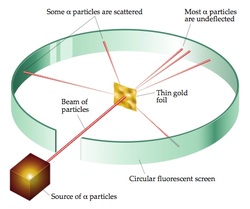 Rutherford's scattering experiment: A stream of alpha particels from a radioactive source was fired at a very thin gold foil. A flash at the fluorescent screen is observed when the alpha particles strikes the screen. The experiment was carried out in a vaccum so this reduces the chance of collision between aplha particles and the air molecules so indeed reduces the chance of losing energy. Conclusions: * Most of fast moving alpha particles went straight through the gold foil. Therefore, the atom is mostly empy space. * Some of the alpha particles were deflected back through significant angles, this suggest that the centre of the atom must be tiny but contain a lot of mass and it was named nucleus by Ernest Rutherford. He was awarded Nobel prize for his discovery. * The alpha particels were repelled, so the nucleus must be postive charge as the charge on the alpha particle is positive so postive- postive repels. * Atoms are overall neutral so the electrons must be on the outside of the atom, separating one atom from the next. 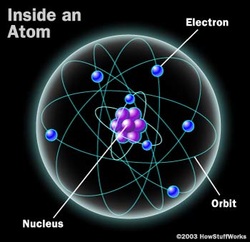 Basic structure of the atom: An atom is made up of nucleus containing protons and neutrons and the nucleus is surrounded by the cloud of electrons. The relative sizes: Atom= 10^-10 Radius of nucleus= 10^-14- 10^-15 Radius of molecule= 10^-10 - 10^-6 Definitions: Nucleon number: A nucleon is a proton or neutron and is also know as mass number. Nuclide: A nuclide is a particular combination of neutrons and protons. |
|
| The World Of Physics |
|
